WHAT IS ALREADY KNOWN ON THIS TOPIC
-
Several tools are available to assess the risk of sudden cardiac death in patients with hypertrophic cardiomyopathy (HCM). Among them, programmed electrical stimulation (PES) is not endorsed by current American and European guidelines, and its role in risk stratification remains controversial due to limited evidence supporting its predictive value.
WHAT THIS STUDY ADDS
-
This systematic review and meta-analysis demonstrate that PES inducibility is strongly associated with a more than 10-times increased risk of major arrhythmic events in patients with HCM. PES shows high sensitivity (81%) and negative predictive value (98%) for identifying at-risk individuals, challenging the current consensus that PES lacks clinical relevance in this setting.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
-
These findings suggest that PES could have a role in refining risk stratification strategies for HCM, particularly for intermediate-risk patients. Further prospective studies are needed to validate its independent predictive value and determine whether PES should be integrated into contemporary risk assessment models.
Introduction
Risk stratification for sudden cardiac death (SCD) in patients with hypertrophic cardiomyopathy (HCM) has evolved over the past several years. The most recent European guidelines have somewhat filled the ‘transatlantic’ gap with the American College of Cardiology (ACC)/American Heart Association (AHA) approach by incorporating additional clinical or imaging high-risk features not included in the clinical risk tool used to estimate the 5-year risk of SCD.1–3 However, the challenge and debate regarding the modalities for accurately identifying individuals at the highest risk of SCD remain. In that respect, the question of incorporating a functional electrophysiological method to complement the actual risk stratification may be raised, especially in patients with myocardial scarring.
Over the years, the use of programmed electrical stimulation (PES) has unequivocally remained either not recommended (Class III), or not considered, by both US and European guidelines. Currently, risk stratification for SCD in HCM is performed entirely in a non-invasive manner.1 2 4 The absence of PES as a risk stratification tool in current guidelines mainly results from its invasive nature and the fact that inducibility is generally considered non-specific.5 However, more recent studies have challenged this paradigm, suggesting that PES may play a role in improving risk stratification for SCD in HCM.6
We conducted this systematic review and meta-analysis in order to assess the actual evidence supporting or opposing the use of PES for risk stratification of major arrhythmic events (MAE) in patients with HCM.
Materials and methods
This systematic review and meta-analysis was registered on the PROSPERO international prospective register of systematic reviews under the ID CRD42024497521. Results are reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 statement (online supplemental table 1).
Literature search
Data were collected from PubMed/MEDLINE, Embase and the Cochrane Library, from inception to 1 February 2024, using the search terms: “hypertrophic cardiomyopathy”, “electrophysiological testing/study”, “programmed ventricular/electrical stimulation” (online supplemental table 2). Two cardiologists (AS and PP) independently conducted the literature review process, with discrepancies resolved by a third reviewer, PM.
Eligibility criteria and selection process
All studies assessing the incidence of SCD, resuscitated cardiac arrest or appropriate implantable cardioverter-defibrillator (ICD) interventions according to PES inducibility in patients with HCM were included. Patients with a history of sustained ventricular arrhythmia or a resuscitated cardiac arrest were excluded. Non-human, non-English and incomplete studies were excluded. Since this analysis used previously published data at the study level, ethical approval was not required.
Quality assessment
The methodological quality of each included study was independently evaluated by two coauthors (AS and PP) using the MINOR (Methodological Index for Nonrandomized Trials) tool for prospective cohort studies. This tool assesses validity through eight criteria for non-comparative studies: clearly stated aim, inclusion of consecutive patients, prospective data collection, appropriate endpoints, unbiased assessment of endpoints, adequate follow-up period, less than 5% loss to follow-up and prospective calculation of study size. For comparative studies, four additional criteria are evaluated: adequate control group, contemporary groups, baseline equivalence of groups and appropriate statistical analyses. Each category is scored from 0 to 2 based on these criteria.7
Data collection and outcomes
Clinical and echocardiographic characteristics related to risk stratification in HCM were systematically collected when available. These included age, sex, family history of sudden death, syncope, ventricular arrhythmia on Holter, maximal wall thickness and left ventricular outflow tract obstruction.
The primary outcome of the study was the incidence of MAE according to inducibility during PES in patients with HCM. MAE was defined as a composite of SCD, appropriate ICD intervention or resuscitated cardiac arrest. Non-sustained ventricular tachycardia (VT) was not considered a positive outcome.
Statistical analyses
Continuous variables were described as means±SD and dichotomous variables as frequencies and percentages. The diagnostic performance of PES was analysed by calculating sensitivity, specificity, as well as positive predictive value (PPV) and negative predictive value (NPV), to assess the test’s ability to predict arrhythmic events. Sensitivity and specificity were calculated from a standard 2×2 contingency table of PES results versus arrhythmic outcomes during follow-up. Specifically, sensitivity was defined as the proportion of patients who experienced an arrhythmic event and tested positive by PES (ie, true positive/(true positive+false negative)), whereas specificity was the proportion of patients who did not experience an arrhythmic event and tested negative by PES (ie, true negative/(true negative+false positive)). We also calculated PPV and NPV from the same table. Meta-analysis was conducted by pooling the results of the published incidence of the predetermined outcomes, using the Mantel-Haenszel random-effects model. ORs and their 95% CIs were used as summary statistics. The percentage of variation across studies due to heterogeneity rather than chance was estimated using the I2 statistic. Heterogeneity was classified as low (<25%), moderate (25–50%) and high (>50%). Finally, a ‘leave‐one‐out’ sensitivity analysis was conducted by sequentially excluding each individual study from the pooled data to assess the robustness of our overall findings. Statistical significance was defined as a p value <0.05. Statistical analyses were performed using RevMan (RevMan, V.5.4.1, The Cochrane Collaboration, Copenhagen, Denmark) and Stata Statistical Software, Release 17.0 (StataCorp. 2021, StataCorp LLC, College Station, Texas, USA).
Results
Article selection
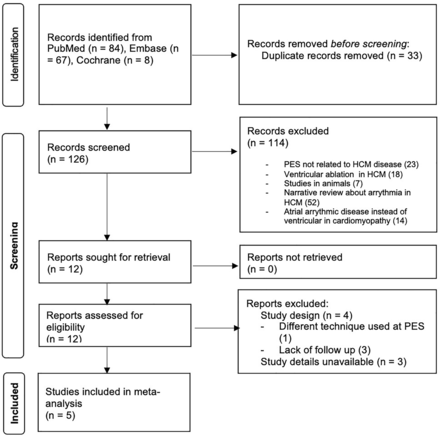
The initial search yielded 159 articles (84 from PubMed, 67 from Embase, 8 from the Cochrane Library). Of these, 126 articles were excluded based on the abstract review. A full-text review of the remaining 12 articles led to the inclusion of 5 studies in the final analysis (figure 1).8–12 Detailed information regarding the included studies is provided in table 1.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow diagram.
Studies evaluating the incidence of arrhythmic events according to inducibility at PES
Characteristics of the overall cohort
A total of 587 patients (ranging from 3 to 203 patients per study, 66% male, age 38±14 years, inclusion period: 1987–2018) were included. Based on the available data, maximal wall thickness exceeding 30 mm was present in 47 cases (14%), left ventricular outflow tract obstruction in 223 (42%), a family history of sudden death in 146 (25%), ventricular arrhythmia on Holter monitoring in 187 (32%) and a history of either syncope or presyncope in 240 (41%). Comparative analysis regarding the prevalence of risk factors composing the HCM-Risk score could not be performed because of incomplete data regarding PES inducibility.
Indications for PES included the presence of clinical symptoms such as palpitations, presyncope and syncope, or the presence of abnormal paraclinical findings, such as the detection of ventricular arrhythmia on Holter monitoring, or the combination of both clinical and paraclinical findings.
PES was performed at both the right ventricular apex and outflow tract in all studies except one.8 All stimulation protocols included up to three extrastimuli (S2, S3, S4).8 Inducibility criteria included sustained monomorphic VT as well as polymorphic VT and ventricular fibrillation (VF). Detailed procedural information is provided in table 2.
Baseline characteristics according to inducibility at PES
Outcomes
Of the 587 patients, 188 (32%) were inducible overall, with monomorphic VT induced in about one-third of cases (36%). The overall incidence of MAE across all 5 studies was 7.2%, ranging from 0% to 24%. After a mean±SD follow-up of 54±42 months, MAE occurred in 18% of inducible patients and in 2% of non-inducible patients (OR 10.83, 95% CI 3.52 to 33.34, p<0.001; figure 2 and online supplemental figure 1). The overall mortality rate was 0.4%. Moderate heterogeneity was observed among studies (I2=27%). Detailed information regarding procedural data and outcomes is provided in table 3.

Pooled incidence of major arrhythmic events according to inducibility at EPS.
Detailed procedural data and outcome
The sensitivity of PES to predict MAE was 81%, and specificity 72%. The PPV was 18%, and the NPV was 98%. The contingency table of MAE according to inducibility at PES is presented in online supplemental table 3.
Quality assessment
The risk of bias in all five included studies was low to moderate based on the MINOR assessment tool (online supplemental table 4). The mean score for our comparative study was 16±4.
Sensitivity analysis
In sensitivity analyses, omitting one study at a time, the pooled OR ranged from 6.56 to 22.11, with CIs that consistently excluded 1, and with I² values ranging from 0% to 50%. No single study significantly altered the effect estimate or the overall conclusions. A summary of this analysis is presented in online supplemental table 5.
Discussion
We conducted a systematic review with meta-analysis to evaluate the value of PES for the risk stratification of MAE in patients with HCM. The major finding of this study is that PES inducibility is indeed predictive of a higher arrhythmic risk in primary prevention. Odds for future MAE were more than 10 times higher among inducible compared with non-inducible patients.
Our findings stand in contradiction with current guidelines and the broad consensus against the use of PES in patients with HCM.1 13 Our explorative analysis therefore suggests that there should be a reappraisal of the contribution of PES in the risk stratification of patients with HCM. Considering the conflicting evidence, even if debatable and with arguable limitations, we believe that PES should not be simply disregarded at this stage as it may play a role in risk stratification, at least in patients at intermediate risk. However, our analysis does not provide information on the independent prognostic value of PES when combined with other classical risk markers, in particular for patients at intermediate HCM risk score, or in grey zones after assessment of AHA/ACC risk factors. Similarly, the cost-effectiveness of incorporating PES would depend on the net reclassification improvement compared with the current risk stratification strategy.
PES has often been criticised for being a non-specific risk stratification tool because of overly ‘aggressive’ stimulation protocols inducing non-specific arrhythmias.4 5 While this limitation is certainly relevant, it must be put in perspective with other well-established high-risk markers. The 18% PPV of PES found in our meta-analysis is indeed comparable to the PPV of typical markers used to stratify risk of sudden death in patients with HCM, such as extensive late gadolinium enhancement (≥15% of left ventricular mass),14 15 the presence of an apical aneurysm15 16 or left ventricular systolic dysfunction15 17 18 (PPV 9–21%, 22–26%, and 16–19%, respectively). Moreover, the excellent negative predictive value of PES at 98% could be especially valuable in an intermediate-risk patient when making decisions regarding SCD prevention. In that respect, it must be noted that the value of these guideline-supported cardiac magnetic resonance-based risk markers has also been demonstrated in patients deemed at low-to-intermediate ESC SCD risk score.14 15
While the majority of SCD in HCM is related to malignant arrhythmias arising from a susceptible electrophysiological substrate, the current risk stratification exclusively relies on non-electrophysiological means. Saumarez et al recently (re)opened the debate on the use of physiologically based electrophysiological methods to predict the arrhythmic risk. In their article, five predictive studies of SCD were analysed to compute receiver operating characteristics. Four studies using traditional clinical and MRI-based risk factors were directly compared with a study using an invasive electrophysiological method, the ‘paced electrogram fractionation analysis’. In brief, this method involves detecting small, fractionated potentials at the end of the paced electrogram that appear as the coupling interval of the extrastimuli is reduced. This method is expected to expose explicitly the electrophysiological effects of disarray that could form a re-entrant electrophysiological substrate.19–22 The electrophysiological method demonstrated a higher predictive capacity and identified 90% of at-risk patients with a 20% false-positive rate (area under the curve (AUC) ≈ 0.89), whereas predictive methods using conventional clinical risk factors and MRI detected only 50–60% of events and yielded an AUC of ≈0.70. Another functional but non-invasive electrophysiological method has recently been reported by Rahimi et al. They assessed QRS fragmentation on surface ECG during sinus rhythm, which may be influenced by conduction heterogeneity much like electrogram fragmentation. They developed a QRS fragmentation metric quantified from a 3-minute high-resolution 12-lead ECG and tested this new metric in patients with HCM with prophylactic ICD and no history of VT/VF. After a mean follow-up of 5 years, this new metric predicted VT/VF in multivariate analysis, whereas non-ECG-based risk factors and scores did not.23 Collectively, these findings further support the potential role of functional electrophysiological markers to complement the current clinical risk stratification.
Functional electrophysiological methods, including standard PES, paced electrogram fractionation analysis or surface ECG fragmentation, are all related to the burden and composition of myocardial scar, which constitutes the electrophysiological substrate.24 Indeed, Francia et al recently correlated the features of inducible ventricular arrhythmia during PES with the scar extent and composition detected by MRI.25 They observed a gradual increase in scar mass and the prevalence of border zone channels among patients, progressing from those who were non-inducible, to those who were inducible but with non-sustained arrhythmia, and finally to those with inducible sustained arrhythmias.25 All patients with inducible monomorphic VT or monomorphic-initiated poymorphic VT/VF had late gadolinium enhancement. These preliminary findings suggest that PES and electrophysiological methods may complement the use of cardiac MRI in a risk stratification strategy. Additional risk assessment with PES could, for instance, be selectively implemented in patients with MRI evidence of a potential arrhythmic substrate.
Our results, however, have limitations related to the quality and heterogeneity of the collected data, which are inherent to the studies included in our meta-analyses. Specifically, data on patients’ characteristics based on inducibility during PES were incompletely reported, and the sample size of some studies was relatively small. Furthermore, details on the follow-up methodology are limited as were the detailed reasons for ICD implantation. More importantly, the patients’ risk profile according to the currently established risk stratification strategy concerning PES inducibility is not known. Accordingly, the incremental value of PES beyond the risk profile determined using traditional risk factors cannot be assessed. Our analysis certainly does not allow us to define the role of PES in a risk stratification strategy incorporating the traditional clinical and MRI factors but aims to stimulate a reassessment of the contribution of PES in the risk stratification of patients with HCM.
Conclusion
Our analysis provides evidence of an association between PES inducibility in patients with HCM and the occurrence of arrhythmic events in primary prevention, with an estimated 10 times higher risk. These findings challenge current guidelines and the prevailing consensus against the use of PES in the context of HCM. Further larger-scale prospective studies should specifically evaluate whether PES may offer an independent incremental value in the context of a contemporary non-invasive risk stratification strategy, particularly for patients classified as being at intermediate risk of SCD.
Data availability statement
Data are available upon reasonable request.
Ethics statements
Patient consent for publication
Ethics approval
Not applicable.