Introduction
The inflammatory response is paramount in the pathogenesis of atherosclerosis.1 Colchicine has broad anti-inflammatory effects that include inhibition of microtubule formation, mitosis, leucocyte motility and cytokine release from a range of inflammatory cells.2 Low-dose colchicine (0.5 mg once daily) has been investigated in multiple randomised clinical trials involving >30 000 patients with atherosclerotic disease. It is associated with a lower risk of major adverse cardiovascular events, in particular, in patients with chronic disease, and treatment is recommended by European and American guidelines.3–5 Clinical trials, however, show conflicting results in acute coronary syndrome and stroke, and heterogeneity in the effect size in the chronic phase, probably pertaining to various biological and methodological phenomena.6–9
Coronary CT angiography (CCTA) is a non-invasive imaging modality that enables quantitative assessment of pericoronary inflammation and coronary atherosclerotic plaque characteristics. Pericoronary inflammation attenuates local intracellular lipid accumulation in preadipocytes in pericoronary adipose tissue (PCAT), changing its composition from a lipid to a more aqueous phase, making it detectable by CCTA.10 PCAT attenuation is an established imaging biomarker of coronary inflammation. Increased PCAT attenuation has been associated with a greater future risk of plaque rupture and acute coronary syndrome.11 12 In addition, particular plaque characteristics, such as a high burden of non-calcified plaque and low-attenuation plaque, have been associated with plaque instability and an increased risk of coronary events.13 Conversely, statin therapy has been shown to promote plaque calcification in invasive and non-invasive imaging studies. This is considered one of the mechanisms by which statins lower the risk of cardiovascular events.14
We hypothesised that treatment with low-dose colchicine in patients with chronic coronary disease would attenuate coronary inflammation, as evidenced by a lower PCAT attenuation, and would stabilise coronary plaques, leading to more favourable plaque morphology, independent of statin treatment.
Methods
Study design and population
This was a prespecified cross-sectional imaging substudy of the international, investigator initiated, randomised, placebo-controlled, Low-Dose Colchicine 2 (LoDoCo2) Trial. The design and primary results of the trial have been previously published.15 In brief, the LoDoCo2 Trial randomised 5522 patients with chronic coronary disease to receive colchicine 0.5 mg daily or matching placebo on a background of optimal medical therapy. Median follow-up was 28.6 months (IQR 20.5 months to 44.4 months). The primary outcome was the composite of cardiovascular death, myocardial infarction, ischaemic stroke or ischaemia-driven coronary revascularisation. In the trial, the risk for the primary outcome was 31% lower in participants allocated to colchicine as compared with placebo. For this substudy, four sites from the Netherlands with an established cardiovascular imaging research infrastructure were selected. For this imaging subanalysis, we included trial patients with multivessel coronary artery disease and a prior acute coronary syndrome who had been clinically stable for at least 6 months. Most important exclusion criteria were prior coronary artery bypass grafting, advanced renal failure (serum creatinine >150 µmol/L or an estimated glomerular filtration rate <50 mL/min), or a history of myositis. Online supplemental figure 1 shows the patient selection and study design.
Coronary CTA acquisition
Participants underwent contrast-enhanced CCTA using a 128-slice ‘Somatom Definition Flash’ scanner (Siemens Healthcare, Forchheim, Germany) in four participating sites in the Netherlands (Alkmaar, Amersfoort, Breda and Heerlen). The scans were performed at the end of the trial just before the close-out visits, while participants were still on study medication and investigators were blinded to treatment allocation. The same protocol, image setting and dose of contrast medium was used at all four centres, which can be found in the extended methods in the online supplemental appendix.
Quantitative assessment of CT
Quantitative assessment of PCAT attenuation and atherosclerotic plaque characteristics was performed using artificial intelligence-enabled plaque analysis software (Autoplaque, V.3.0, Cedars-Sinai Medical Center) by expert readers (AL, JK, PM, KG) in the core laboratory at Cedars-Sinai Medical Center, who were blinded to clinical data. PCAT attenuation was measured in the proximal segments of all three major epicardial coronary arteries: the left anterior descending artery, the left circumflex artery and the right coronary artery, as previously described.12 For the left anterior descending artery and the left circumflex artery, the proximal 40 mm was analysed, excluding the left main given its variable length and possible absence. For the right coronary artery, the proximal 10–50 mm was analysed, excluding the first 10 mm to avoid effects of the aortic wall. For scans performed at 100 kV, PCAT attenuation was divided by a conversion factor of 1.11485 to be comparable to scans performed at 120 kV.10 12
In coronary arteries with a stent in the proximal segment, PCAT was also analysed along the length of the stent. Coronary CTA scans with suboptimal image quality due to noise or artefacts were excluded.
PCAT was defined as adipose tissue located within a radial distance from the outer coronary artery wall equal to the average diameter of the respective vessel. PCAT attenuation was calculated as the mean attenuation of all voxels between −190 Hounsfield units (HU) to −30 HU within this volume of interest, around the proximal segments of all three major epicardial coronary vessels on a per-participant level.16
Quantitative assessment of atherosclerotic plaque characteristics was performed using axial and multiplanar reconstructed CCTA images. All coronary segments with diameter ≥2 mm were evaluated according to an 18-segment model.16 Specifics can be found in the extended methods in the online supplemental appendix.
Clinical data and laboratory assessments
All clinical data were collected at randomisation and captured in electronic case report forms. For the purpose of this analysis, statin dose was converted to atorvastatin dose equivalent, in which atorvastatin dosages of 40 mg and 80 mg were considered high-intensity statin therapy.
Blood samples for laboratory data on inflammatory biomarkers were collected on the day of the CCTA study, or as close as possible. Information on the assays can be found in the extended methods in the online supplemental appendix.
Outcomes
The primary outcome was the between-group difference in PCAT attenuation. The exploratory secondary outcomes were differences in plaque morphology (volumes and burden of non-calcified, calcified plaque, low-attenuation and dense calcium plaque) and inflammatory biomarkers (high sensitivity C-Reactive protein (hs-CRP) and interleukin 6 (IL-6)).
Statistical analysis
Central tendencies and distribution of continuous parameters are displayed using means with SD or medians with IQR (25th and 75th percentiles) in case of normally and non-normally distributed variables, respectively. For the non-normally distributed biomarkers, we used the geometrical mean.
Normality was assessed visually using histograms and Q-Q plots. Categorical variables are presented as counts and percentages.
The study was designed to have 90% power to detect an absolute difference of 3.8 HU in PCAT attenuation, assuming a median PCAT attenuation of −84.2 HU in the experimental arm and −80.7 HU in the control arm, with an SD of 6.5 HU.17 This yielded a necessary sample size of 146 patients. To account for participant withdrawal or inadequate image quality, we aimed to recruit 170 participants.
To assess the direction and effect size of differences between treatment groups for the primary outcome, mixed models and multivariable generalised linear regression models were used. A value of p<0.05 was regarded as statistically significant. The reported p values of the secondary outcomes are for hypothesis-generating purposes, as we did not correct for multiplicity. Additional information on the modelling and statistical methods can be found in the extended methods in the online supplemental appendix.
Results
Participants
In total, 153 of 172 participants approached for the imaging substudy underwent CCTA. Sixteen patients withdrew consent, two had intercurrent illness and one had a known contrast allergy. Two scans with inadequate image quality were excluded, resulting in 151 studies for the final analysis (online supplemental figure 1)
The characteristics of participants were equally distributed between treatment arms and are shown in table 1. The mean age of the participants was 64.4 years (SD±8.0) and 14% were female. All participants were on antiplatelet and/or anticoagulant therapy, and almost all were on statin therapy (91%), with half of participants receiving high-intensity statins (57%). All patients in both groups had undergone prior coronary stenting at study enrolment. The average low-density lipoprotein cholesterol level was 2.2 mmol/L (SD±0.9, equals 85.1 mg/dL, SD±34.8). The median duration of exposure to trial medication at the time of CCTA was 28.0 months (IQR 23.9 months to 33.6 months) in the colchicine group and 28.3 months (IQR 23.6 months to 33.2 months) in the placebo group.
Baseline characteristics
PCAT attenuation and plaque morphology
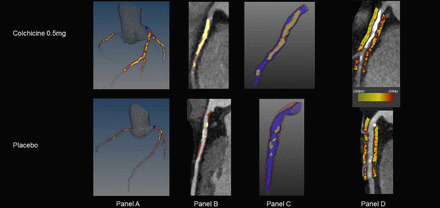
Figure 1 shows representative case examples of quantitative plaque and PCAT analysis. The primary and secondary outcomes are shown in table 2
figure 2 and figure 3. The median PCAT attenuation did not differ between the two treatment groups: −79.5 HU (IQR −85.0 HU to −72.5 HU) in the colchicine group versus −78.7 HU (IQR −84.1 HU to −72.1 HU) in the placebo group (adjusted difference −1.0, 95% CI −2.7 HU to 0.7 HU, p=0.236). Results were similar after exclusion of the stented segments within each vessel: median PCAT attenuation −77.2 HU (IQR −84.0 HU to −72.4 HU) in the colchicine group versus −75.6 HU (IQR −82.4 HU to −70.1 HU) in the placebo group (adjusted difference −1.3, 95% CI −3.9 HU to 1.3 HU, p=0.335).
Representative case examples of quantitative plaque and PCAT analysis in the left anterior descending artery for two patients in the Low-Dose Colchicine 2 (LoDoCo2) Trial, stratified by treatment. (A) Patient-specific three-dimensional reconstruction of the coronary tree; (B, C) artificial intelligence-enabled plaque analysis with calcified plaque in yellow overlay and non-calcified plaque in red overlay; (D) PCAT attenuation around the proximal 40 mm of the vessel.

Differences in plaque composition according to treatment arm. The black centre line denotes the median value (50th percentile), while the box contains the 25th to 75th percentiles. The black whiskers mark the 5th and 95th percentiles, and values beyond these upper and lower bounds are outliers, marked with black dots.

Plaque composition. The figure shows a stacked bar chart depicting the proportion of calcified plaque (red) and non-calcified plaque (blue) per treatment arm.
Pericoronary adipose tissue (PCAT) attenuation and plaque morphology
Non-calcified plaque volume did not differ between the treatment groups (colchicine: 552.2 mm3, IQR 271.0 mm3 to 942.5 mm3; placebo: 493.3 mm3, IQR 326.4 mm3 to 819.3 mm3; adjusted difference 52.3 mm3, 95% CI −70.9 mm3 to 175.43 mm3, p=0.407). Calcified plaque volume, however, was significantly higher in the colchicine group compared with the placebo group (169.6 mm3, IQR 65.1 mm3 to 334.5 mm3 vs 113.1 mm3, IQR 49.0 mm3 to 241.6 mm3, adjusted difference 59.0 mm3, 95% CI 3.1 mm3 to 115.0 mm3, p=0.041, figure 3). This difference persisted when calcified plaque volume was indexed to the total vessel volume, with a calcified plaque burden in the colchicine group of 9.6% (IQR 5.1 % to 14.8%) compared with 7.0% (IQR 4.0% to 12.0%) in the placebo group (adjusted difference 2.4%, 95% CI 0.2% to 4.7%, p=0.035), and also when indexed to total plaque volume (adjusted difference 4.5%, 95% CI 0.4% to 8.6%, p=0.034, figure 2). Dense calcified plaque volume was higher in the colchicine group (192.8 mm3, IQR 55.9 mm3 to 394.4 mm3) versus the placebo group (144.3 mm3, IQR 53.7 mm3 to 264.3 mm3) with an adjusted difference of 61.5 mm3 (95% CI 0.9 mm3 to 122.0 mm3, p=0.048). Volumes and burdens of low-attenuation plaque did not differ between the overall treatment groups.
PCAT attenuation in relation to inflammatory biomarkers and treatment duration
We observed no differences in the levels of inflammatory biomarkers between treatment groups: median hs-CRP was 1.12 mg/L (IQR 0.64 mg/L to 2.04 mg/L) in the colchicine group as compared with 1.14 mg/L (IQR 0.64 mg/L to 2.04 mg/L) in the placebo group (p=0.247) and median IL-6 was 1.96 ng/L (IQR 1.47 mg/L to 3.43 mg/L) in the colchicine group as compared with 2.20 ng/L (IQR 1.46 mg/L to 3.55 mg/L) in the placebo group (p=0.164). No correlation was found between hs-CRP levels and PCAT attenuation in either treatment group: for colchicine, Spearman’s r=−0.061 (p=0.468), for placebo, Spearman’s r=0.099 (p=0.276). Also, no correlation was found between IL-6 levels and PCAT attenuation (for colchicine r=−0.05, p=0.549, for placebo r=0.028, p=0.762) (online supplemental figure 2). There was also no correlation between duration of exposure to low-dose colchicine and PCAT attenuation (r=−0.109, p=0.340), or duration of exposure to low-dose colchicine and total calcified plaque burden (r=−0.038, p=0.741) (online supplemental figure 3)
PCAT attenuation and plaque morphology in relation to high-intensity statin use
Although PCAT attenuation appeared to be lower in the colchicine group as compared with the placebo group in patients on low-intensity statins as compared with high-intensity statins, no significant interaction was found (online supplemental figure 4).
The higher median calcified plaque volume and burden in the colchicine group as compared with the placebo group occurred irrespective of the intensity of statin therapy (pinteraction=0.225). Among participants on low-intensity statin therapy, colchicine was associated with a significantly lower low-attenuation plaque burden versus placebo, while this was not observed in those on high-intensity statin therapy: median low-attenuation plaque burden was 4.8% for colchicine versus 6.7% for placebo in low-intensity statin users as compared with 4.7% for colchicine versus 4.5% for placebo in high-intensity statin users (pinteraction=0.037) (online supplemental figure 5).
Discussion
This prespecified LoDoCo2 substudy used a cross-sectional, nationwide, design to investigate coronary inflammation and plaque morphology in 151 patients with chronic coronary disease after 2 years of exposure to low-dose colchicine or placebo. Our main findings were: (1) There was no significant difference in PCAT attenuation between participants on low-dose colchicine versus placebo, (2) Calcified plaque volume and burden was higher in participants on low-dose colchicine, independently of statin intensity, and (3) In participants using low-intensity statin therapy, the burden of low attenuation plaque was lower in participants on low-dose colchicine than in participants on placebo.
Non-invasive assessment of the processes that drive atherosclerotic plaque progression and rupture has recently gained interest.18–20 PCAT attenuation has emerged as a patient-level risk marker of future acute coronary syndrome.12 21 Further, PCAT attenuation has been used to track changes in the coronary inflammatory status in response to systemic anti-inflammatory treatments.22 In view of these promising results, we sought to assess the effects of low-dose colchicine on coronary inflammation, using PCAT attenuation as an indirect imaging biomarker. To our knowledge, ours is the first CCTA study to investigate the effects of colchicine on pericoronary inflammation and plaque morphology in chronic coronary disease, with a major strength being the randomised, placebo-controlled design. Although the direction of the difference in PCAT attenuation was concordant with our hypothesis, the effect size was small and not significant. This finding might indicate that colchicine does not change pericoronary inflammation, or changes in patients with chronic coronary are smaller than anticipated. Our findings are in line with preliminary evidence from other investigators demonstrating no temporal changes in PCAT attenuation following 12 months of treatment with low-dose colchicine.23 Our study had a substantial proportion (42%) of coronaries with stents in proximal vessel segments, which may have affected the CT attenuation of the adjacent PCAT due to partial volume averaging. This is consistent with our observation that the estimated difference in PCAT attenuation was more pronounced when excluding stented segments. Lastly, inherent to the design of the current study, no pretreatment CT scans were available, which precluded correction for baseline PCAT attenuation and temporal assessment of within-patient changes in PCAT attenuation. The median PCAT attenuation values we measured were comparable to those reported in other studies involving patients with chronic coronary disease, but 10% lower than the cut-off values used to define high-risk populations.12 However, comparing absolute PCAT attenuation values between studies still is challenging due to variations caused by CT scanners, scan parameters and processing algorithms between studies.21 24
Few imaging studies have examined the effect of colchicine on markers of plaque stability. Yu et al used intracoronary optical coherence tomography to assess fibrous cap thickness during an acute coronary syndrome and after 12 months in patients randomised to low-dose colchicine or placebo. They showed a greater increase in fibrous cap thickness in patients with colchicine as compared with placebo: minimal fibrous cap thickness change in the low-dose colchicine group was 87.2 μm (95% CI 69.9 µm to 104.5 µm) vs 51.9 μm (95% CI 32.8 µm to 71.0 µm) in the placebo group, suggesting a plaque stabilising effect.25
To date, the only non-invasive evaluation of colchicine and changes in coronary plaque characteristics comes from Vaidya et al, who performed a non-randomised, single-centre, observational study in which 80 patients received either optimal medical treatment or optimal medical treatment and colchicine 0.5 mg once daily after an acute coronary syndrome. Colchicine was associated with a significantly reduced low-attenuation plaque volume as measured by serial CCTA over 12 months. Mean low-attenuation plaque volume in the low-dose colchicine group changed from 38.8 mm3 at baseline to 23.0 mm3 at 12 months (mean change 15.9 mm3 (−40.9%)) versus from 39.0 mm3 at baseline to 32.3 mm3 at 12 months (mean change 6.6 mm3 (−17.0%), value of p<0.001) in those not receiving colchicine).26 No significant temporal changes were observed in calcified plaque volume. In contrast, our study used a randomised and placebo-controlled design, in a population with higher median low-attenuation plaque volumes. Differences in low-attenuation plaque volume between treatment groups in our study occurred only in those on low-intensity statins. Our study also revealed higher calcified plaque volumes and burden in participants treated with colchicine, and also dense calcified plaque volume (defined by a CT attenuation >1000 HU) was higher. These findings should be interpreted with caution, as they were explorative and serve a hypothesis-generating purpose. Histopathological studies have shown that lesions with dense calcium are more likely to be stable plaques, exhibiting sheet-like (>3 mm) calcification and a lower burden of necrotic core.27 Furthermore, recent CCTA studies have shown that high-density calcified plaque is associated with reduced risk for future coronary events. In addition, rates of transformation to high-density calcified plaques are greater in the presence of statin therapy.28 29
These findings may help in patient selection when choosing anti-inflammatory treatment. The causal role of inflammation in atherosclerosis has been confirmed via Mendelian randomisation studies and clinical outcomes trials with several anti-inflammatory drugs.30 31 However, it remains challenging to identify which patients benefit the most. The observational evidence of the role of inflammation in cerebrovascular disease has not yet been translated to therapeutic strategies, and studies investigating colchicine in patients with acute myocardial infarction show conflicting results.7 32–34 Whether the high inflammatory status in acute myocardial infarction explains the lack of treatment effect in the Colchicine and Spironolactone in Patients with Myocardial Infarction (CLEAR) Trial is not sure, since a late treatment effect neither arose in this trial. The absence of clinical effect in that trial is also in contrast with the changes in plaque morphology observed in the study of Vaidya et al, in which treatment was also commenced after myocardial infarction.26 Inflammation stratified analyses from the Canakinumab Anti-Inflammatory Thrombosis Outcomes Study showed a marked differential response in treatment effect in benefit of patients reaching below median IL-6 levels.35 Inflammation stratified analyses and plaque subgroup analyses for CLEAR will help understand these differences. Earlier safety concerns on increases in infections or non-cardiovascular mortality were, however, not confirmed by recent studies. Nevertheless, treatment initiation should be done with caution in patients with mild-to-moderate renal impairment, and treatment should be withheld in those with renal or liver failure.36 37
Our study has several limitations. First, we conducted a cross-sectional analysis at the end of treatment, without baseline or sequential imaging studies. This precludes correction for baseline differences in plaque burden and characteristics and PCAT attenuation between groups and limits temporal assessment of the effects of colchicine on plaque progression or pericoronary inflammation. The ongoing Effect of Colchicine on Progression of Known Coronary Atherosclerosis in Patients with STable CoROnary Artery Disease CoMpared to Placebo Trial (n=84) will provide additional insights on this.38 Assessment at the end of treatment in our study could also introduce non-random differences between the groups due to treatment effects or survival bias. However, since fatal events were rare in the main trial and prior non-fatal events were no exclusion for the imaging substudy, we believe that the risk of observational bias due to differential censoring is low.
Second, the representation of women was not proportional to the prevalence of cardiovascular disease among women in the general population.39 Female patients develop manifest atherosclerosis at older age and exhibit distinct phenotypes of atherosclerosis due to differences in lifetime exposure to risk factors.40 This may limit the interpretation and generalisability of our results and remains a matter of concern, since results from clinical trials are translated into guideline recommendations that apply equally to women and men.41
Third, we did not have supplementary imaging modalities, such as positron emission tomography or invasive plaque imaging (eg, intravascular ultrasound or optical coherence tomography) or intracoronary blood sampling for cytokines to corroborate our findings on CCTA.