Good morning, and welcome to day two of Cancer Research Catalyst’s live coverage of the AACR Annual Meeting 2026. Once again, we will be sharing updates throughout the day, including more coverage of the Educational Program, the AACR Runners for Research 5K Run/Walk, the start of the clinical trials program, the Discovery Science Plenary, and much more.
If you missed our live updates from yesterday, catch up on what happened on Friday.
Today’s Coverage
(All times are Pacific Time.)
10 p.m. – Discovery Science Plenary Session Explored Minimal Residual Disease and the Role of Cell Plasticity in Solid Tumors
The concept of minimal residual disease (MRD), which was first formulated for blood cancers, is now being extended to solid tumors to uncover hidden disease and refine treatment decisions.
The first Plenary Session of the AACR Annual Meeting 2026, titled “The Next Frontier in Minimal Residual Disease: Solid Tumors,” explored the latest research on the evolution of drug resistance, the role of cancer cell heterogeneity and plasticity, and novel strategies to detect and target residual disease.
The session was chaired by Maximilian Diehn, MD, PhD, of Stanford University. While MRD has been used to personalize treatment in hematologic malignancies for decades, it is now an area of growing interest in solid tumors thanks to advances in our understanding of the biology of MRD and our ability to detect it, said Diehn.
He pointed out that there are two types of MRD in solid tumors: MRD after systemic therapy, which occurs in patients with advanced disease and is due to persister cells, and MRD after local surgery in patients with early-stage disease, due to microscopic metastases.
The first presenter, Aaron N. Hata, MD, PhD, of Massachusetts General Hospital, reviewed mechanisms underlying the evolution from drug-tolerant persistent cells to acquired resistance to targeted therapies and disease relapse in lung cancer, focusing on the role of the mutator enzyme APOBEC3A, whose expression and activity are induced during treatment, in the acquisition of genomic instability.
He also presented emerging evidence deriving from the study of residual disease in ALK-positive lung cancer and supporting the involvement of lineage-constrained lung epithelial injury states. This model could provide a useful paradigm for studying and targeting the early stages of therapy resistance, Hata said.
Jean-Christophe Marine, PhD, of the VIB-KU Leuven Center for Cancer Biology in Belgium, focused on therapeutic strategies to address cancer cell heterogeneity and plasticity. “Today I will flip the script a bit and I will show you that cancer plasticity can actually be leveraged to our advantage in order to mitigate therapy resistance,” said Marine, suggesting that, by promoting tumor heterogeneity, therapy also creates vulnerabilities that can be identified and targeted.
Marine discussed an innovative approach called Origami that his team has devised, a physics-based framework that leverages cell morphology by measuring cell surface and volume to monitor phenotypic diversity and plasticity.
Presenter Dan A. Landau, MD, PhD, of Weill Cornell Medical College and the New York Genome Center, said that one mission of his team is to generate novel single-cell multi-omics technologies that can study different aspects together at the level of the single cell. Another parameter they are trying to bring into the picture is time: They use phylogenetics as a “temporal microscope” to study genetic and phenotypic evolution and understand whether the different cell states and phenotypes associated with residual disease are heritable. To do so, the team generates high-resolution phylogenetic trees directly from patient samples.
Another focus of Landau’s research is the development of new methods to use the cancer genome as a dynamic clinical sensor to read out residual disease and predict outcomes. The current paradigm to do this, he explained, is to use deep targeted sequencing because ctDNA is very sparse, but there are only a few thousands genomic equivalents in a blood sample, Landau said, which creates a limitation he described as “input ceiling.” He suggested whole genome mutational integration as a strategy to overcome input ceiling.
Last presenter Jeanne Tie, MD, of Peter MacCallum Cancer Centre and the Walter and Eliza Hall Institute in Australia, discussed the impact of MRD in guiding clinical trial design for colorectal cancer. Tie emphasized how patient selection for adjuvant chemotherapy trials is currently guided by risk and not by the presence of disease, which results in treating many patients—exposing most of them to unnecessary toxicities—to benefit a few. On the other hand, a trial design based on treating MRD-positive disease would allow for enrichment of patients who will benefit from therapy, translating to a dramatic improvement in trial efficiency, Tie said.
She reviewed the potential of circulating tumor DNA (ctDNA) as a clinical tool for patient selection based on MRD status, thanks to technological progress that dramatically improved assay sensitivity. According to Tie, ctDNA response holds great promise as an endpoint, but its regulatory acceptance for solid tumors will require a coordinated global effort to validate the correlation between ctDNA reduction and disease-free survival.
8:10 p.m. – The First-ever Group of AACR-ONF Oncology Nurse Scholars
Oncology nurses play an integral role in clinical research and cancer care, and AACR has long been committed to recognizing, supporting, and elevating their contributions through its enduring partnership with the Oncology Nursing Society (ONS) and the Oncology Nurse Foundation (ONF). This year, the organizations launched the AACR-ONF Oncology Nurse Scholars Program through which the following 10 oncology nurses were selected to attend the Annual Meeting and take part in a curated educational experience:
- Amy Ballou, DNP, MSN, RN, APRN, NP, PhD candidate, of University of South Carolina
- Maria Berkeley, BSN, RN, of Community Memorial Hospital
- Cameron Herbst, MSN, RN, of University of Pittsburgh
- Brianna Kammerer, MSN, RN, APRN, NP, of The University of Texas (UT) MD Anderson Cancer Center
- Nilesh Kalariya, PhD, MSN, RN, APRN, NP, of UT MD Anderson Cancer Center
- Hyein Kim, MSN, RN, of Emory University
- Madeline Merrill, MSN, RN, APRN, NP, of Memorial Sloan Kettering Cancer Center
- Kim Navarrete, MSN, RN, APRN, NP, of UT MD Anderson Cancer Center
- Peg Rosenzweig, PhD, CRNP, AOCNP, of University of Pittsburgh
- Monica Wagner, PhD, RN, of Case Western Reserve University
This afternoon the nurse scholars participated in a mentorship and networking roundtable during which they were able to engage with prominent leaders in the cancer research community including but not limited to AACR CEO Margaret Foti, PhD, MD (hc); AACR President Lillian Siu, MD, FAACR, of Princess Margaret Cancer Centre; AACR Immediate Past President Patricia M. LoRusso, DO, PhD (hc), FAACR, of Yale Cancer Center; Jedd D. Wolchok, MD, PhD, FAACR, of Weill Cornell Medicine; Jayesh Desai, MBBS, of Peter MacCallum Cancer Centre; Matthew G. Vander Heiden, MD, PhD, of Massachusetts Institute of Technology (MIT) and the Koch Institute; Elizabeth Platz, ScD, MPH, of Johns Hopkins Bloomberg School of Public Health; and Jill Feldman, a lung cancer patient and advocate.
The nurse scholars were paired with researchers by different areas of focus, including translation research and precision oncology; drug development and clinical trials, data science, biomarkers and AI; health disparities, population science and survivorship; immunotherapy, cellular therapy and precision oncology; and basic science, tumor biology and metastasis.
“They have PhDs. They may have a doctorate in nursing,” said Platz. “They are exactly the same as the basic scientists, translational scientists and population scientists who are doing the research. They are the ones asking the questions, getting the funding and leading the research, developing the team, but they didn’t know that this meeting applied to them.”
Siu, LoRusso, and Foti then provided presentations to encourage engagement and collaboration between the nurse scholars and AACR.
Throughout the meeting, the nurse scholars have been invited to attend a selection of scientific sessions relevant to nursing practice and patient care while also exposing them to the full spectrum of cancer research.
Stay tuned for a blog post after the meeting to learn more about the program and its impact on the first-ever selection of AACR-ONF Oncology Nurse Scholars.

7:35 p.m. – Undergraduate Student Caucus and Poster Competition Announces Winning Posters
The undergraduates assembled at the AACR Undergraduate Student Caucus today were on tenterhooks this afternoon, and for good reason. Having presented their original research work (at the AACR Annual Meeting, no less), the students waited to see who among them would receive the prestigious poster awards from the Margaret Foti Foundation.
But before the dramatic moment came, Program Chair Paul Mischel, MD, FAACR, had some words of wisdom for the eager young faces. He began by asking for a show of hands in the room for those whose lives had been touched by cancer. Dozens of hands were raised, and Mischel nodded.
“It matters to you, doesn’t it? That’s why you’re here. It matters for me, too,” he said.
Mischel told them the story of how, as a young man, he watched his father die of gastric cancer. The experience, painful though it was, would set Mischel on the path to medicine and, ultimately, cancer research. “I became a pathologist to look the enemy in the eyes,” he said, and pathology was just the start of a journey that led him to his definitive work in extrachromosomal DNA’s role in cancer.
Mischel encouraged the scientists-to-be to delve into the uncharted waters of unanswered research questions with the same enthusiasm that had brought them to the Annual Meeting. The future of cancer research, he stressed, was very much in their hands.
“You are a part of us now,” he said. “We are delighted to have you here.”
Next to the stage was AACR President Lillian Siu, MD, FAACR, who welcomed the students with her own fond memories of starting out as a young researcher.
“You are our next generation,” she said. “Many years ago, I sat in your seat. … You’re hungry for science, you’re hungry for learning—you are going to be the stars, and you are the future.”
Siu then laid down a challenge for the undergraduates: to approach at least 10 people they didn’t know and question them—about their research, their background, and their motivations. That, she said, was the key to forming successful networks.
When AACR CEO Margaret Foti, PhD, MD (hc), ascended the rostrum, she thanked them for their participation and encouraged all the students to make the most of the Annual Meeting, and she celebrated the brightness of the futures on display.
Whether they would go on to work in academia or industry, the undergraduates were destined to be the next generation of trailblazers so long as they remained dedicated, she said. Foti also had a request: “When you go back home,” she said, “brag about what you just enjoyed!”
Foti then announced this year’s winners. Two $700 third-place awards were named: Lori Khatchaturian, for “HDAC1-specific inhibition increases MALAT1 expression in myeloid-derived suppressor cells and breast cancer cells and induces cell cycle arrest”; and Durga Meenakshi Panneerdoss, for “LIPA inhibition enhances the therapeutic efficacy of DNA-damaging agents in ovarian cancer through induction of ER stress and enhancing DNA damage.”
Then came the two $1,000 second-place prizes: Khandoker Ifaz Ahmed, for “Keap1 loss promotes cancer stemness and anti–PD-1 resistance in head and neck cancer”; and Ahmad Ziada, for “Dual remodeling of RCC-associated fibroblasts via miR-196b antagomir and COUP-TFII inhibitor ultrasmall lactoferrin-lipid nano-assemblies augments immunotherapy.”
Finally, after it seemed as though everyone had heard a drumroll in their head, the grand $1,500 prizewinner was named: Maryam Faisal, for “Targeting Yap1-B7-H3 signaling stimulates CD8⁺ antitumor response preferentially in male SHH medulloblastoma.”
The five meritorious young scientists took their place beside Foti for a celebratory photograph as they held aloft their well-deserved, oversized checks. They will also be presenting their posters again during the Poster Session, “2026 AACR Undergraduate Scholars and Margaret Foti Undergraduate Prize Recipients,” from 9 a.m. to 12 p.m. on Monday in the Exhibit Hall.

7:05 p.m. – Team Science Award Recognizes the Cancer Dependency Map Team
The Broad Institute Cancer Dependency Map (DepMap) Team was recognized with the 2026 AACR Team Science Award for their novel work in systematically mapping genetic dependencies across cancer cells. The team created a comprehensive resource that reveals genes and pathways essential for tumor survival. By combining large-scale CRISPR functional genomic screens, drug response data, and multiomic profiling, the DepMap team has uncovered lineage- and genotype-specific cancer vulnerabilities, including synthetic lethal dependencies such as WRN in microsatellite instability cancers and PRMT5 dependencies in cancers harboring MTAP deletions. Their work has transformed the discovery and prioritization of therapeutic targets and has become a foundational resource for cancer research and drug development worldwide.
The team members include Francisca Vazquez, PhD (team leader); Jesse S. Boehm, PhD; Catarina Campbell, PhD; Glenn Cowley, PhD; Joshua M. Dempster, PhD; Levi A. Garraway, MD, PhD, FAACR; Todd R. Golub, MD, FAACR; William C. Hahn, MD, PhD; James McFarland, PhD; Philip Montgomery, MS; David E. Root, PhD; Jennifer A. Roth, MBA, MA; William R. Sellers, MD, FAACR; Yuen-Yi “Moony” Tseng, PhD; Aviad Tsherniak, MS; and Barbara A. Weir, PhD.

“DepMap is a remarkable consortium of academic scientists, data scientists, and health care professionals who pioneered systematic mapping of genetic dependency across cancer cells,” said session chair Amina Zoubeidi, PhD, of the University of British Columbia in Canada, while presenting the award. “Their work has reshaped our understanding of cancer biology and opened new avenues in the discovery of precision therapies.”
Accepting the award on behalf of the team, Vasquez said that at the beginning of the journey, when the team only had ideas and concepts, the team shared the conviction that building a dataset like this was going to be worthwhile. “We thought it would matter, we just didn’t know how much,” she said. “So being here, recognized by AACR in this room with scientists and clinicians both from industry and academia who are working to turn discoveries into therapies, means more than I can express because this is the community for which we built DepMap.”
During her lecture, Vazquez reviewed the history of DepMap and its major milestones; the technological advancements that enabled its evolution; the computational infrastructure that was required; and the partnerships involved. She also acknowledged all the people whose work behind the scenes has been instrumental for the growth and success of DepMap.
Beyond the scientific advancement that the platform has facilitated, DepMap has democratized discovery, said Vasquez, as she reflected on the enormous impact that this resource has had on the scientific community: The DepMap portal has more than 11,000 users per week; the public release of data (26 scheduled releases so far) has allowed laboratories from 186 countries over the last year, regardless of their funding availability and computational expertise, to use the data to make their discoveries; and DepMap’s data releases are independent of academic publication, to ensure that the data are readily available to the community.
In his address, Sellers acknowledged that the DepMap consortium would not exist without support from industry funders. He reviewed some of the next steps for the platform and some of the questions that remain to be addressed. “We are not there yet,” he said. The next frontier includes the integration of an artificial intelligence-enabled interface to make data easier to explore, analyze, and visualize by people who are not computational biologists.
6:30 p.m. – Clinical Trial Results: The Predictive Value of ctDNA After Neoadjuvant Immune Checkpoint Inhibition
The second Clinical Trials Minisymposium of today focused on advances for perioperative treatments, with two presentations examining how circulating tumor DNA (ctDNA) levels after neoadjuvant immunotherapy may help predict treatment outcomes.
New results from a sub-study of the NSABP B-59/GBG-96-GeparDouze trial, presented by Marija Balic, MD, PhD, MBA, of the University of Pittsburgh and the NSABP Foundation, showed that the presence of ctDNA after completion of neoadjuvant therapy, but before surgery, was associated with residual invasive disease and a more than 10-fold higher risk of distant recurrence among patients with early-stage triple-negative breast cancer. The trial is evaluating the impact of adding atezolizumab (Tecentriq) to neoadjuvant therapy, followed by surgery and adjuvant atezolizumab. The new findings reported here build upon results from the trial presented at the 2025 San Antonio Breast Cancer Symposium, which showed that ctDNA positivity after surgery was associated with elevated risk of recurrence.
Results from the CheckMate 77T trial, which is evaluating neoadjuvant chemotherapy with either perioperative nivolumab (Opdivo) or placebo in patients with resectable non-small cell lung cancer, also showed that the presence of ctDNA after neoadjuvant therapy, but before surgery, may adversely impact patient outcomes. Patients receiving perioperative nivolumab had higher event-free survival than those receiving perioperative placebo regardless of their ctDNA status after neoadjuvant therapy. However, the magnitude of nivolumab’s impact varied by ctDNA status: after a median follow-up of 41 months, nivolumab treatment was associated with a 52% higher rate of event-free survival among those who were ctDNA-negative after neoadjuvant therapy, but this increase dropped to 24% among patients with detectable ctDNA after neoadjuvant therapy.
The findings support the efficacy of perioperative nivolumab for treating resectable NSCLC and highlight ctDNA-based biomarkers as clinically meaningful tools for risk stratification, said presenter Tina Cascone, MD, PhD, of The University of Texas MD Anderson Cancer Center.
6 p.m. – Take a Sneak Peek of the Exhibit Hall
Exhibitors are currently hard at work setting up the Exhibit Hall. Opening tomorrow at noon, the Exhibit Hall will house numerous poster sessions on wide-ranging topics, as well as the Patient Advocate Pavilion and hundreds of exhibitor booths!
Stop by anytime during these hours:
- Sunday: 12–5 p.m.
- Monday: 9 a.m.–5 p.m.
- Tuesday: 9 a.m.–5 p.m.
- Wednesday: 9 a.m.–12 p.m.



5:30 p.m. – Applying Precision Techniques to Childhood Cancer
An Educational Session held this morning, “Small Patients, Big Problems: Precision Medicine Approaches for Childhood Cancer,” covered the myriad ways in which personalized approaches (many of which have originated from research on adult cancer patients) can be brought to bear in the pediatric clinic.
Session chair Sarah K. Tasian, MD, of the Children’s Hospital of Philadelphia, emphasized the importance of using precision approaches to drive progress beyond the already impressive improvements achieved in the treatment of many pediatric patients.
Illustrating the benefits of precision pediatric cancer approaches, the first speaker, David Ziegler, MBBS, MD, PhD, of the Children’s Cancer Institute in Australia, reviewed the impact that broad-based integration of precision methods with pediatric oncology can have on patient outcomes.
Such improvements in treatment, he said, depend on leveraging precision therapies against known targets, but he pointed out that targeting requires continuous refinement due to the complexity of tumor signaling. He illustrated this point with a picture that he likened to a tumor’s inner workings: a snarl of tangled wires and cables.
Methods like DNA methylation profiling and next-generation sequencing, Ziegler emphasized, unlock a level of insight into tumor biology that has already improved outcomes for pediatric tumors, including the highest-risk tumors.
One study he cited from the Zero Childhood Cancer Program showed that 71.4% of pediatric cancer patients had therapeutic targets identified by whole-genome sequencing and RNA sequencing—and 5.2% even had a change in their diagnosis. Even patients with lower-risk tumors, said Ziegler, can benefit from precision approaches.
“It’s not just about the ‘magic bullet,’ but also about the diagnostic tests,” said Ziegler.

Katherine A. Janeway, MD, MMSc, of Dana-Farber Cancer Institute, applied a similar line of thinking to sarcomas—a subtype of cancers that, according to Janeway, represents an area of stalled progress compared with other indications. The lack of progress in sarcomas, she said, has been due to a lack of established biologic subtypes (itself partially a problem of physical constraint, due to the physical difficulty of working with bone, according to Janeway). Researchers have recently begun to turn to AI-driven approaches to overcome the difficulties in classifying pediatric sarcomas—a step toward better-informed treatment guidance and tumor analysis.
But establishing biologic subtypes for osteosarcoma in pediatric and adolescent and young adult (AYA) populations offers an opportunity to apply targeted approaches to an indication that, she said, has not been receptive to new anticancer developments. She reviewed some of her work that endeavored to gather both tumor and patient data to create such subtypes.
Elevated MYC expression and MYC amplification, Janeway said, have been shown to predict worse overall survival in pediatric and AYA osteosarcoma patients—identifying MYC as a possible target for personalized cancer treatment.
Janeway also talked about targetable modifications in Ewing sarcoma using molecular profiling, and she proffered a framework for developing Ewing sarcoma risk groups. She pointed to the identification of STAG2 mutations as an outcome-associated target in patients with Ewing sarcoma from the Children’s Oncology Group cohort. She called for further research and validation, and she expressed enthusiasm for future work to develop actionable biomarkers.
The session concluded with a presentation from Tasian about precision oncology’s application to pediatric leukemias.
The widespread adoption of risk-adapted chemotherapy has led to great success in treating children with acute lymphoblastic leukemia (ALL), but Tasian stressed that one size does not fit all. The genetic heterogeneity occurring across both B- and T-cell pediatric ALL populations indicates a need for precision analyses and therapies, she said.
As an example of an ongoing precision approach to pediatric ALL, Tasian pointed to investigations of ALL that, despite not testing positive for the actionable Philadelphia chromosome (the first-known cancer fusion gene), nevertheless have clinical characteristics resembling Philadelphia-positive ALL. These ALL variants, she said, are termed “Philadelphia-like ALL” for their similarity. Though themselves a heterogenous family of tumors, Philadelphia-like ALL tumors can be broken down into therapeutically actionable subgroups.
Additionally, Tasian covered how childhood cases of acute myeloid leukemia (AML) are also complicated by genetic heterogeneity, particularly when such tumors develop chemoresistance. These AML relapses are very difficult to salvage, Tasian said, stressing a need for more investigations. Encouragingly, however, clinical trials targeting several aspects of chemoresistant pediatric AML are ongoing, she said, and she pointed to FLT3 inhibitors for pediatric FLT3-mutant AML as a potential “gamechanger.”
4:55 p.m. – The Next Frontier in Proximity-Inducing Therapies
Organized in association with the AACR Chemistry in Cancer Research Working Group, today’s four-part Chemistry to the Clinic series delved into next-level conjugates, proximity-inducing therapies, novel therapeutic targets and modalities, and new frontiers in drug discovery from transcript to protein.
One of the four sessions, “Chemistry to the Clinic Part 2 of 4: Beyond Inhibition—The Next Frontier in Proximity-inducing Therapies,” examined various proximity-based approaches, as well as a new strategy for functional screening of compounds.
Proximity-inducing therapies bring together a target protein and a cellular mediator to exert various effects, such as degradation, stabilization, phosphorylation, or relocalization of the target, explained William J. Gibson, MD, PhD, of Dana-Farber Cancer Institute. Proximity-inducers can also be used to redirect transcription factors to promote expression of apoptotic genes or to disrupt the function of an essential cellular protein. Gibson noted that proximity-inducing therapies are rapidly advancing, as seen by the recently reported success of the proximity-induced degrader daraxonrasib for pancreatic cancer. He also shared data from an investigational compound designed to bridge Y220C-mutated p53 with PLK1 to prevent PLK1 from carrying out its essential functions.
Session chair Michael A. Erb, PhD, of The Scripps Research Institute, discussed an investigational proximity-inducing compound called PCIP-1 that can phenocopy the anticancer effects of PARP inhibitors in BRCA-deficient cells. The compound bridges PARP and BRD4 to induce PARP trapping onto DNA, leading to DNA repair inhibition and cell death, even in cells with acquired resistance to conventional PARP inhibitors, Erb noted.
Steven Banik, PhD, of Stanford University, shared a new high-throughput screening approach for functional testing of candidate proximity-inducing molecules. The screening method is inspired by the natural cellular response to protein degradation, which is transcription activation to produce more of that protein. The researchers designed their system to tie transcription activation of a particular gene to a reporter, such as GFP. In a high-throughput screen, detection of the reporter would serve as a readout that the target protein was degraded by the candidate compound. He added that RNA barcoding could also be used as a reporter, essentially enabling “proteomics by sequencing.”
4:25 p.m. – Communication Tips for Creating Buzz About Research
“Even the most brilliant science is invisible … if others don’t understand,” said Kristen Dahlgren, of Parker Institute for Cancer Immunotherapy, during the Educational Session “From Bench to Buzz: Making Your Research Resonate for More Funding and Attention.” During the session, Dahlgren and her fellow panelists offered tips on how researchers can create more awareness about their research.
Dahlgren, a former journalist and cancer survivor, spoke from experience when suggesting that researchers need to relay the heart or emotion about their research to grab the attention of the public or even potential funders. Since most patients don’t want to read through academic papers—and most likely don’t have access to them—scientists instead need to focus the story on why the research matters to patients. How can it help them? What is the gut punch that may shock them? One of the best ways to do this is to tell the story through the eyes of the patient. She added it is important to share the person beyond their disease so others can relate and empathize with them.

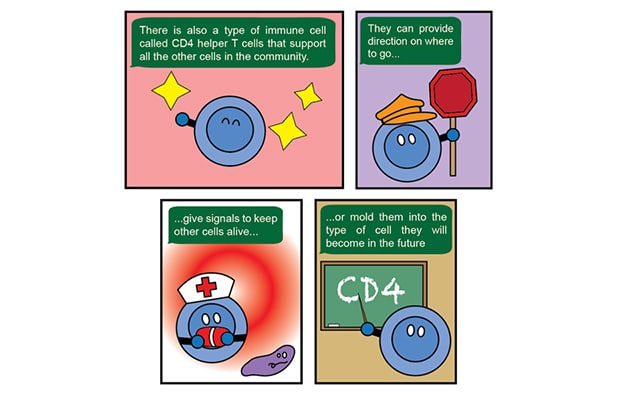
Jaye Gardiner, PhD, of Tufts University, next offered advice on how researchers can better translate their science to the public. She suggested following the Pixar method of storytelling, which involves starting with the “once upon a time” or the basic backstory behind the research, then explaining the problem the research is trying to solve, how this research could change the status quo, the steps necessary to lead to this change, the climax of the research (which is the most important finding), and the future implications. And like Pixar, Gardiner also suggests using visuals to tell this story.
“You don’t have to be good at art,” she emphasized. Gardiner, who makes her own science-based comics, offered the example below to show how researchers can use very simple shapes and visual metaphors of everyday life to explain a concept like CD4 cells.
Finally, with so many people now turning to artificial intelligence (AI) bots like ChatGPT for answers to health questions, cancer survivor and AI enthusiast Saranne Rothberg, of The ComedyCures Foundation, provided insights into how researchers can amplify their messages by ensuring their work is being included in AI-generated responses. As she explained, one of the top things AI bots look for when deciding what information to share is FAQs. She suggested researchers create online FAQs about what they want AI to know about themselves and their work. She also said researchers should be “cross-pollinating,” meaning partnering with researchers outside of their institutions to share each other’s work as that will also make it more authoritative in the eyes of AI and hence more likely to be picked up by the bots.
2:20 p.m. – Clinical Trial Results: Long-term Updates on Approved Immunotherapies
With a record number of clinical trial abstracts—265—this year’s clinical trials program has been expanded to include five Clinical Trials Minisymposia, starting with two today.
“The expanded number of Minisymposia will provide attendees insights into exciting developments across the spectrum of clinical research, from early-phase results of novel therapeutic strategies, a session focused on biomarker-driven approaches, to long-term updates on approved treatments,” Jayesh Desai, MBBS, of Peter MacCallum Cancer Centre and a cochair of this year’s Annual Meeting Clinical Trials Committee, explained to AACR Annual Meeting News.
Among the trials presented at this morning’s session, “Updates in Anticancer Immunotherapies,” were two of those long-term updates on approved treatments. Atezolizumab (Tecentriq) was approved for patients with alveolar soft part sarcoma (ASPS)—an ultra-rare cancer with incidence rates highest among 15- to 35-year-olds—in December 2022 based on early results from an ongoing phase II clinical trial. As of June 2025, 54 patients have been enrolled in the trial with the duration of atezolizumab response ranging between 10 and 69 months.
“Because many patients experienced durable responses and remained on immunotherapy for several years, we wanted to understand the potential long-term effects of this treatment, particularly for adolescents and young adults with ASPS who may receive immunotherapy for extended periods,” Alice P. Chen, MD, of the National Cancer Institute (NCI), explained in an AACR press release about the study.

Seventeen patients—between the ages of 11 and 56 with a median age of 29—received either atezolizumab or atezolizumab followed by atezolizumab plus the vascular endothelial growth factor (VEGF) inhibitor bevacizumab (Avastin) for more than two years. Of these patients, one who was receiving atezolizumab plus bevacizumab experienced grade 3 aspartate aminotransferase (AST), which can be a sign of liver damage; another who was receiving the combination therapy experienced grade 2 hypertension—or high blood pressure—and grade 2 proteinuria, which can be a sign of kidney damage; and one who was receiving atezolizumab monotherapy experienced grade 2 pruritus, which is an itchy feeling causing one to scratch their skin.
“Among patients who received immunotherapy for longer than two years, we did not observe evidence of increased toxicity and this finding held even among patients treated for more than five years,” Chen said. “This is especially important for adolescents and young adults with ASPS, who may live for many years with metastatic disease.”
Further, Chen explained that none of the atezolizumab-related TRAEs resulted in individuals discontinuing with the study and all the symptoms were eventually resolved.
In another presentation, Anna Maria Di Giacomo, PhD, MD, of the University of Siena in Italy, shared 10-year survival data from the phase III NIBIT-M2 study examining treatment with fotemustine, fotemustine and ipilimumab (Yervoy), or ipilimumab and nivolumab (Opdivo) for patients with melanoma who have asymptomatic brain metastases. From January 2013 to September 2018, 23 patients were treated with fotemustine, 26 were treated with fotemustine and ipilimumab, and 27 were treated with ipilimumab and nivolumab.
After a median follow-up of 125 months, patients in the ipilimumab and nivolumab arm had a higher median overall survival (29.2 months), 10-year overall survival rate (31.2%), and intracranial progression-free survival (28.8%), compared with those in the fotemustine arm (8.5 months, 13%, and 4.3%, respectively) and the fotemustine and ipilimumab arm (8.2 months, 7.7%, and 7.7%, respectively).
Further, Di Giacomo explained how tumor fraction and a tumor-specific methylation score (T-meth Score) calculated using cell-free DNA may serve as biomarkers to predict long-term survival. Patients were stratified at baseline based on median tumor fraction and T-meth Scores, and those with tumor fraction and a T-meth Score below median values had significantly higher median overall survival.
12:45 p.m. – Meet With Potential Employers at the Career Fair
The AACR Cancer and Biomedical Research Career Fair—a premier recruiting event for scientists at all career levels—is still going on in Hall G of the convention center. Job seekers can meet with exhibitors from academia, government, and industry, including:
- Cancer Research UK
- Cancer Science Institute of Singapore, National University of Singapore
- Fulgent Genetics
- Moffitt Cancer Center
- National Cancer Institute
- Pacific Permanente Group
- Provider Solutions & Development by Providence
- Rutgers Cancer Institute
- School of Biomedical Sciences, Faculty of Medicine, The Chinese University of Hong Kong
- Science Careers
- St. Jude Children’s Research Hospital
- The University of Texas (UT) MD Anderson Cancer Center
- UT Southwestern Simmons Comprehensive Cancer Center and Graduate School
- American Association for Cancer Research
Additionally, anyone who creates a new profile on CancerCareers.org is entered to win one of three prizes: an Apple iPad, a $250 AMEX gift card, and a $150 AMEX gift card.
The Career Fair will end at 3 p.m., so be sure to stop by before then.

12:15 p.m. – Applying Spatial Multiomics to Study Neural-immune Circuits
During this morning’s Educational Session on “Mapping Neural-Immune Circuits: Spatial Multiomics in the Tumor Microenvironment,” speakers examined spatial technologies and the ways they are being applied to understand the roles of neural and immune cells in the tumor microenvironment.
“The tumor microenvironment is now understood to be a deeply neural-immune ecosystem. Neural and immune cells don’t operate in isolation but interact in ways that meaningfully shape cancer progression and response to therapy,” said session chair Amit Moran, MD, PhD, of The University of Texas MD Anderson Cancer Center, in an interview with Cancer Research Catalyst.
He added that the goal of the session was to give researchers practical insights into spatial multiomics so that they can interrogate these neural-immune circuits in their own research programs.
To that end, Logan Walsh, PhD, of McGill University, kicked off the session by giving attendees a primer on spatial multiomics technologies, as well as considerations for platform selection, tissue handling, and image analysis when using these tools. He explained that spatial multiomics allow researchers to understand how variables such as gene expression, protein expression, epigenetic modifications, and metabolic alterations vary across the three-dimensional space of the tumor microenvironment. When analyzed over time, these tools can help capture the changes that occur as a tumor develops, evolves, and progresses.
Currently available technologies use fluorophores, positional barcodes, or metal tags in conjunction with microscopy, sequencing, or mass spectrometry to map various types of omics data—which can then be used to identify where cells are located (a process known as segmentation), and to determine each cell’s phenotype or activation state, Walsh explained.
William Hwang, MD, PhD, of Massachusetts General Hospital and Harvard Medical School, then discussed how these technologies have been applied to better understand neural-immune circuits within tumors. As one example, he shared how he and colleagues used spatial transcriptomics to identify mediators of perineural invasion. By mapping cell type-specific gene expression at different distances from an invading nerve, they found that PDGFD signaling was enriched in the cancer cells near the nerve and that this signaling promoted neuronal outgrowth and perineural invasion. They also examined how the neural and immune-cell composition of cellular neighborhoods impacted the expression of tumor-promoting genes.
Lastly, Amit offered insights into how to translate spatial multiomics data into clinical impact. By comparing spatial multiomics data from patients whose tumors responded to the immune checkpoint inhibitor cemiplimab (Libtayo) with data from patients whose tumors did not respond, he and colleagues identified neural and immunological factors, such as perineural invasion, that influence a tumor’s likelihood of response.
“Integrating spatial transcriptomic and proteomic data with clinical outcomes and population-level datasets … can transform mechanistic observations into clinically actionable insights,” he said.
11:45 a.m. – AACR Runners for Research 5K Run/Walk Brings 1,100+ Runners, Exceeds Fundraising Goal
It was a beautiful morning here in San Diego, California, where the famously picturesque harbor was abustle with a sight familiar to Annual Meeting attendees: the AACR Runners for Research. This year, the sold-out(!) race featured more than 1,100 participants from 31 countries.
The annual AACR Runners for Research 5K Run/Walk (AACR 5K) is a cherished tradition of the AACR Annual Meeting, and the more than $250,000 raised for cancer is a testament to the enthusiasm and dedication of the participants—some of whom traveled from the other side of the globe to be here and still found the energy for an early-morning 5K.
We had the chance to catch up with some of the runners we spoke with for our preview story (you can learn more about their reasons for running in our preview post). See our interview below with Annie W.Y. Chai, PhD, a scientist representing Cancer Research Malaysia and a former AACR Global Scholar-in-Training Award recipient.
Industry was well-represented too—including a veritable battalion from Revolution Medicines, who were Visionary Supporters of the race this year. As they were photographed, they let out what you might call their trademark cheer: “Daraxonrasib!”

As the runners warmed up with calisthenics and team cheers, an AACR who’s who delivered a resounding pep talk. AACR Annual Meeting 2026 Program Chair Alice T. Shaw, MD, PhD, FAACR, thanked the crowd for their commitment to funding cancer research and pledged her own support as a fellow runner. Research funding of the kind being raised at the AACR 5K, she said, is absolutely essential to keep the state of the cancer research field advancing while accelerating breakthroughs. “We are all gathered to unite and help fund cancer research—and fund progress!”
AACR President-Elect Keith Flaherty, MD, FAACR, was up next at the mic. “I had my knee replaced last year, and I’m going to break it in today!” he said, eliciting both laughter and cheers. He added, “Every step you take on the course brings us one step closer to a world without cancer.”
Program Chair Paul Mischel, MD, FAACR, could not participate due to a conflicting morning session (“I’m dressed for running!” he joked, wearing a full suit), but he offered his sincere gratitude to the runners.
“This run says a lot. There has never been a more important time for cancer research … By being here today, you are sending a critical message: that cancer research matters, and that we’re in this together,” said Mischel.

When the starting gun fired, the front line of runners took off faster than you can say “plenary,” as they raced down the bayfront in a blur of green and black, running with the wind that blew in from the Pacific. Wave after wave of runners headed down the track, many in their Runners for Research finery but some sporting their own costumes—including superhero capes and neon-green mohawks.
Some members from the AACR Board of Directors could be spotted as the runners surged forth, having swapped their conference formalwear for running gear. Their team, The Cure Catalysts, included notables like the 2026-2027 AACR President-Elect Robert H. Vonderheide, MD, DPhil, FAACR, Shaw, and former AACR President, Antoni Ribas, MD, PhD, FAACR (who achieved his goal of finishing the race at about 21 minutes).
The festivities lasted for nearly an hour, but it took all of 16 minutes and 49 seconds for the winner to cross the finish line. Keith Bailey, PhD—a field application scientist manager from LUMICKS—won, in addition to his AACR medal, unquestionable bragging rights for finishing the race with a blistering pace of 5 minutes, 28 seconds per mile.
“We [at LUMICKS] have been coming to the AACR Annual Meeting for many years, but it’s been six years since we ran the 5K, so this is a big deal for us,” said Bailey.
And what excites Bailey about the Annual Meeting (other than winning the 5K)? “I love being exposed to and being in discussions with the top minds in cancer research in the world,” he said. “It excites me every year to come to all the research sessions here. And it’s great to meet all the runners that I got to run with today.”

6:05 am – AACR Project GENIE Will Show Off New Harmonization Data Model
Since it launched in November 2015, AACR Project GENIE (Genomics Evidence Neoplasia Information Exchange) has grown into the largest, publicly accessible cancer registry with real-world genomic and clinical data. At a Methods Workshop this morning, attendees can learn about the latest resource to come from this project—the GENIE Data Model (GDM). This is a public, open‑access, scalable framework that helps cancer centers collect and organize clinical and genomic information in the same way so it can be compared and combined more easily.
“When you want to do precision oncology research, you have to gather data from multiple institutions, and you cannot get away with whatever local definitions one institution has created,” explained Jeremy L. Warner, MD, of Brown University and the chair of the Methods Workshop. “If you want to have a common language—describing the extent of disease, whether a drug works, what outcomes matter—all these things that have to be standardized, that’s the backdrop of this session.”
The Methods Workshop, which takes place from 8 to 9:30 a.m. in Room 31 in the upper level of the convention center, will walk attendees through how GDM is structured, what it takes to put it into practice, and the available support for tracking patients over time and bringing together multiple data types.
Learn more about the session and other sessions related to AACR Project GENIE occurring throughout the meeting in this article published in AACR Annual Meeting News.